- - animals:
- - type:
- anti inflammatories
- - usage:
- injectable sterile solution
- - manufacturer:
- Rooyan Darou
- - Packaging:
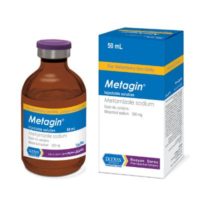
- 50 ml vials
Product properties
Composition
Each ml contains 40mg of tolfenamic Acid.
Mechanism of Action:
Tolfenamic Acid is a non-steroidal anti-inflammatory drug (NSAID) belonging to the fenamate group. Tolfenamic Acid exerts anti-inflammatory, analgesic and antipyretic activities. The anti inflammatory activity of Tolfenamic Acid is mainly due to an inhibition of cyclooxygenase and thus to a reduction in the synthesis of prostaglandins and thromboxanes, which are important inflammatory mediators .Tolfenamic Acid is distributed in all the organs with a high concentration in the plasma, digestive tract, liver, lungs and kidneys. However, the concentration in the brain is low. Tolfenamic Acid and its metabolites do not cross the placental barrier to any great extent. Tolfenamic Acid undergoes extensive enterohepatic recirculation and, as a result, prolonged concentrations are found in plasma.
The elimination half life varies from 8-15 hours in cattle. In cattle, Tolfenamic Acid is eliminated mainly unchanged in faeces (~30%) and urine (~70%).
Indications of use
Treatment and control of musculoskeletal pains and any kind of inflammations in other organs.
As an adjunct in the treatment of acute mastitis, used in conjunction with antibacterial therapy.
As an aid in the control of acute inflammation associated with bacterial respiratory disease, used in conjunction with antibacterial therapy.
Dosage and route of administration
The general dose of product is 4mg/kg of bodyweight ( 1ml/10kg of B.W.).
For use in bovine mastitis, the recommended dosage is 4mg/kg bodyweight (1ml per 10kg bodyweight) as a single IV injection.
For use in bovine respiratory disease, the recommended dosage is 2mg/kg bodyweight (1ml per 20kg bodyweight) by IV or SC injection.
Treatment may be repeated once after 48 hours. If a second injection is needed this should be given in the opposite side of the animal’s body.
In Dogs & Cats
For relief of acute and chronic pains the dose is 4 mg/kg once daily SC, IM of PO for 3-5 days. The injection is suggested for the first dose only.
Contraindication
Usage is contra-indicated in animals suffering from cardiac, hepatic or renal disorders where there is the possibility of gastro-intestinal ulceration or bleeding, or where there is evidence of hypersensitivity to the product.
Drug interactions:
Transient swelling and inflammation can occur at the injection site.
Withdrawal Periods
Milk: 24 hours, milk from the first milking after treatment should be discarded.
Meat: (Cattle) Following intravenous administration :3 days.
Following subcutaneous administration: 7 days
Adverse reactions
Do not sell without veterinary’s prescription. Do not exceed the stated dosage and duration of treatment.
Use aseptic precautions when administering the product. Do not administer other NSAIDs concurrently of within 24 hours of each other. Take care to avoid self-injection. In case of eye or skin contact, wash immediately with water. Do not exceed 20ml per injection site. Following withdrawal of the first dose, the product should be used within 28 days.
Storage conditions
Do not store above 25°C. Keep in dry place and protect from direct sunlight.